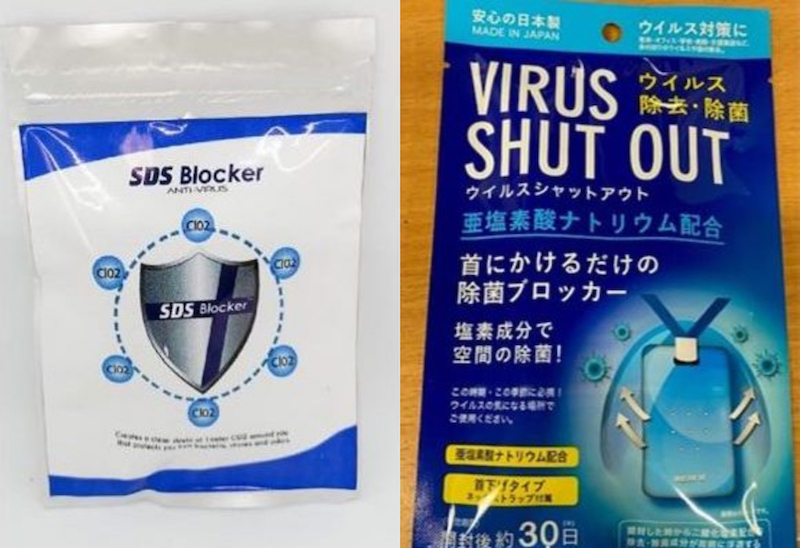
On May 20, the Food and Drug Administration (FDA) issued an advisory against disinfectant products claiming to reduce the risk of contracting COVID-19. Their advisory warned against the use of products like the “Virus Shut-out” and “SDS Blocker Anti-Virus,” which are marketed as protection devices against viruses and bacteria.
FDA Advisory No. 2020-852 || Public Health Warning Against the Purchase and Use of Disinfectants/Biocides Products with…
Posted by Food and Drug Administration Philippines on Tuesday, May 19, 2020
Their advisory advised people to be vigilant about deceptive advertising, since these products have not been proven to cure or kill any viruses or diseases and should not be promoted as such.
FDA also noted that these products contain chlorine dioxide or chlorite, which can cause irritation in the mouth, esophagus and stomach as well as other respiratory problems when inhaled or exposed through skin contact. These products are usually worn around the neck or placed in shirt pockets.
The advisory also encouraged people to follow public health advice from the Department of Health and the World Health Organization, emphasizing that there are currently no treatments for COVID-19.
Header photo courtesy of the Food and Drug Administration – Philippines
Get more stories like this by subscribing to our weekly newsletter here.
Read more:
Duterte warns the public of another lockdown if COVID-19 cases continue to rise
Beware of faulty PPEs: How to ensure quality of locally-produced protective suits
DOH secretary Duque said PH “actually” already at second wave of COVID-19
Writer: ANGELA PATRICIA SUACILLO