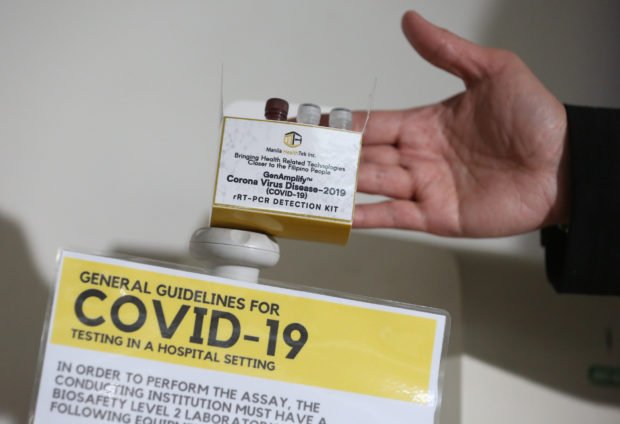
Department of Health (DOH) undersecretary Maria Rosario Vergeire announced in an online media forum on July 10 that the COVID-19 test kit developed by the University of the Philippines (UP) is ready for its relaunch.
“Nakapagsubmit na sila ng bago, na-validate na ng Research Institute for Tropical Medicine (RITM), (at) tinutulungan na natin sila ngayon para maumpisahan na natin ang paggamit sa piling laboratoryo dito sa ating bansa,” said Vergeire.
Called the GenAmplify™ Corona Virus 2019 (COVID-19) rRT PCR Detection kit, the kits are developed by the UP-National Institutes of Health and are manufactured by Manila HealthTek. Each kit is estimated to cost P1,320, which is significantly cheaper than the foreign kits which cost P8,500.
Vergeire also added that the kits have already secured a special certification from the Food and Drug Administration. With this, laboratories only need a final advisory from DOH before they can start using the kits.
According to the Health undersecretary, laboratories have to undergo training for the proper usage of the new test kits first and take a proficiency test as well.
Earlier in May, the GenAmplify™ Corona Virus 2019 (COVID-19) rRT PCR Detection kits were recalled by its manufacturer because 30 percent of the test kits were unable to determine whether the coronavirus is present in the specimen submitted or not. Because of this, 30 percent of the samples had to undergo a repeat test.
Further investigation showed that the imported reagent used in the test kits was “contaminated.” In response, the manufacturer recalled the kits and ordered a new reagent.
Aside from being the first locally made and developed PCR-based COVID-19 test kit approved by the FDA, the GenAmplify™ Corona Virus 2019 (COVID-19) rRT PCR Detection kits are also funded by the Department of Science and Technology.
Header photo from Inquirer.net
Get more stories like this by subscribing to our weekly newsletter here.
Read more:
OVP purchases 10,000 test kits developed by UP scientists
UPNIH testing kits for COVID-19 set to be used starting next week
UP experts create computer systems to connect COVID-19 patients to their loved ones
Writer: YANN MAGCAMIT